Pill look up 316912/14/2023  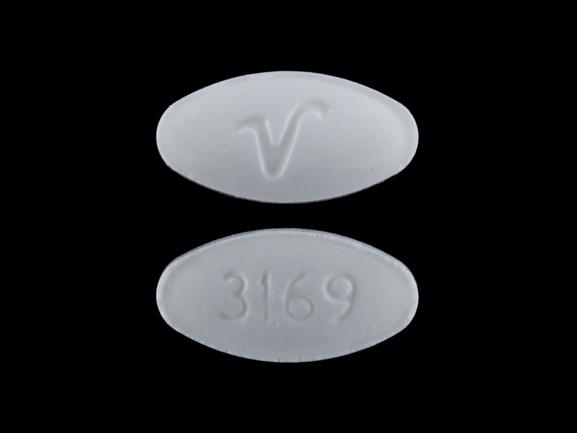
Supplemental potassium chloride and, if required, an aldosterone antagonist are helpful in preventing hypokalemia and metabolic alkalosis. Sudden alterations of fluid and electrolyte balance in patients with cirrhosis may precipitate hepatic coma therefore, strict observation is necessary during the period of diuresis. In hepatic coma and in states of electrolyte depletion, therapy should not be instituted until the basic condition is improved. In patients with hepatic cirrhosis and ascites, furosemide therapy is best initiated in the hospital. There are no significant differences between the two oral formulations in the amount of unchanged drug excreted in urine. Significantly more furosemide is excreted in urine following the IV injection than after the tablet or oral solution. The terminal half-life of furosemide is approximately 2 hours. Peak plasma concentrations increase with increasing dose but times-to-peak do not differ among doses. Although furosemide is more rapidly absorbed from the oral solution (50 minutes) than from the tablet (87 minutes), peak plasma levels and area under the plasma concentration-time curves do not differ significantly. In fasted normal men, the mean bioavailability of furosemide from furosemide tablets and furosemide oral solution is 64% and 60%, respectively, of that from an intravenous injection of the drug. The duration of diuretic effect is 6 to 8 hours. The peak effect occurs within the first or second hour. The onset of diuresis following oral administration is within 1 hour. The unbound fraction averages 2.3 to 4.1% at therapeutic concentrations. Plasma concentrations ranging from 1 to 400 mcg/mL are 91 to 99% bound in healthy individuals. Furosemide is extensively bound to plasma proteins, mainly to albumin. Recent evidence suggests that furosemide glucuronide is the only or at least the major biotransformation product of furosemide in man. The action on the distal tubule is independent of any inhibitory effect on carbonic anhydrase and aldosterone. The high degree of efficacy is largely due to the unique site of action. It has been demonstrated that furosemide inhibits primarily the absorption of sodium and chloride not only in the proximal and distal tubules but also in the loop of Henle. Tell your healthcare provider right away if you get symptoms of gallbladder problems, which may include pain in your upper stomach (abdomen), fever, yellowing of skin or eyes (jaundice), clay-colored stools.Investigations into the mode of action of furosemide have utilized micropuncture studies in rats, stop flow experiments in dogs and various clearance studies in both humans and experimental animals. Gallbladder problems have happened in some people who take Trulicity. Tell your healthcare provider if you have changes in your eyesight (vision) during treatment with Trulicity. Trulicity may cause stomach problems, which could be severe.Ĭhanges in vision. This may cause kidney problems to get worse. In people who have kidney problems, diarrhea, nausea, and vomiting may cause a loss of fluids (dehydration). Stop using Trulicity and get medical help right away if you have any symptoms of a serious allergic reaction which may include swelling of your face, lips, tongue or throat, problems breathing or swallowing, severe rash or itching, fainting, or feeling dizzy, or very rapid heartbeat.Īcute kidney injury. 
Signs and symptoms of low blood sugar may include dizziness or light-headedness, confusion or drowsiness, headache, blurred vision, slurred speech, fast heartbeat, sweating, hunger, shakiness, feeling jittery, weakness, anxiety, irritability, or mood changes. Your risk for getting low blood sugar may be higher if you use TRULICITY with another medicine that can cause low blood sugar, such as a sulfonylurea or insulin. You may feel the pain from your abdomen to your back. Stop using Trulicity and call your healthcare provider right away if you have severe pain in your stomach area (abdomen), with or without vomiting, that will not go away. If you have any of these symptoms, tell your healthcare provider.ĭo not use Trulicity if you or any of your family have ever had a type of thyroid cancer called medullary thyroid carcinoma (MTC).ĭo not use Trulicity if you have Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).ĭo not use Trulicity if you are allergic to dulaglutide or other ingredients in Trulicity.Īsk your healthcare provider how to recognize possible serious side effects and what to do : Watch for possible symptoms, such as a lump or swelling in the neck, trouble swallowing, hoarseness, or shortness of breath. Trulicity may cause tumors in the thyroid, including thyroid cancer. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
